The ability to identify and control individual brain cells (neurones) has generated huge interest in recent years, it's made for some startling discoveries and some incredible images (like the one below). But we're really only beginning to scratch the surface of the potential these technologies hold, and the techniques here are quite controversial.
The actual methods for controlling individual neurones require a lot of complex genetic manipulation, but the general concepts are easier to get to grasps with. The main method used for the genetic manipulation of animal genomes is the cre-lox system. I won't go into the details (you can read a great review by its inventor here) but this technique allows scientists to insert or remove any gene you want, in any cell, even at specific times (when the cells are still growing or mature for example).
The next question many people then asked was, what if I can use this technique to influence an animal's behaviour? Specifically with neurones, could I activate or inhibit the electrical communication between neurones in real time in a real life behaving animal to study the effects of individual sets of neurones on behaviour e.g. learning, memory, reflexes, social interaction, etc?
Turns out, yes you can! The most well known and fashionable technique at the moment is called optogenetics, but there are a couple of other interesting methods, which I'll explain below. The general idea is to insert a gene into the neurones in order to make them sensitive to a stimulus which there are no (or very few) cells that are sensitive to it in the brain and is also able to manipulate or monitor the electrical activity of the neurone (these are almost always ion channels, see here for how ion channels cause neurones to 'fire').
Optogenetics
As you may guess, this involves using optics ie light. By inserting genes for a light sensitive protein that can activate or silence neuronal signalling activity into neurones and applying a light stimulus by using tiny fibre-optic cables, you can control electro-chemical communication between the cells at millisecond time scales.
While this technique offers incredible time resolution, however it's incredibly invasive (you have to insert the fibre-optic fibres into the brain of the animal) and incredibly difficult to get right (you need to make sure the fibres are actually lighting up the neurones you want them to). This creates a huge problem if you want to target neurones deep within the brain, as you need access to those incredibly tricky to reach parts of the brain.
Chemogenetics
Another cool way scientists have been able to manipulate the brains of animals using genetics have involved the use of chemicals, or toxins to be more specific. Researchers have used the Cre-lox system to insert the gene for various toxins eg a fragment of the diptheria toxin into specific neurones in order to kill those cells and observe the effects this has on the mice. Although a bit crude, this can help to identify the brain regions involved in all different types of behaviours, and could help us understand how the brain reacts to and compensates for the loss of specific regions of the brain eg after stroke, dementia, trauma.
Electrogenetics
This technique doesn't necessarily allow researchers to directly manipulate neurones, but it does allow us to watch the activity of specific cells in real time. By inserting genes that express proteins that emit a flourescent light when they sense changes in voltage (indicating electric activity in the neurone) we can easily see each neurone light up individually and trace which other neurones are activated or de-activated by that neurone.
This video below used this technique to show how individual neurones in the auditory cortex are firing in response to sound.
Along the same lines, you can insert specific genes across the entire brain that randomly express a different ratio of red, green and blue fluorescent proteins. That means that each individual neurone has a unique colour, which gives rise to the incredible 'brainbow' images seen above.
Magnetogenetics
This very new method of manipulating neurones involves inserting a gene for an ion channel protein that is activated by magnetic fields you can instantly turn neurones on or off or block anything else from activating it (ie other neurones) while the animal is relatively free to move around and act as normal, without needing a bunch of fibre optic cables delicately positioned in their brains. The fact you don't need to worry about exact positioning of any equipment means that much deeper brain regions or much more specific neurones can be manipulated than with optogenetics. There may also be potential for more analogue control of activity by using a weaker magnetic field that only slightly affects the activity of the neurones being manipulated. However some neurones are already sensitive to magnetic fields, birds and other animals can use them to navigate by, for example.
Ultrasound-ogenetics?
Probably better named audiogenetics (although we can't hear ultrasound...), there is also some very early research into making specific neurones sensitive to ultrasound. This would have many of the benefits of magnetogenetics and both could be the future of genetics based neuroscientific research in animals.
Clearly, there are a number of new and emerging ways in which researchers may be able to investigate the role of very specific neurones in the brain. While some may be initially put off by the idea of manipulating the brains of animals, by understanding these very specific roles, we may eventually reach a more complete understanding of how the brain works and how to fix it when it goes wrong.
 |
| This is a real image of individual neurones and the connections they make, all with individual colourings |
The next question many people then asked was, what if I can use this technique to influence an animal's behaviour? Specifically with neurones, could I activate or inhibit the electrical communication between neurones in real time in a real life behaving animal to study the effects of individual sets of neurones on behaviour e.g. learning, memory, reflexes, social interaction, etc?
Turns out, yes you can! The most well known and fashionable technique at the moment is called optogenetics, but there are a couple of other interesting methods, which I'll explain below. The general idea is to insert a gene into the neurones in order to make them sensitive to a stimulus which there are no (or very few) cells that are sensitive to it in the brain and is also able to manipulate or monitor the electrical activity of the neurone (these are almost always ion channels, see here for how ion channels cause neurones to 'fire').
Optogenetics
As you may guess, this involves using optics ie light. By inserting genes for a light sensitive protein that can activate or silence neuronal signalling activity into neurones and applying a light stimulus by using tiny fibre-optic cables, you can control electro-chemical communication between the cells at millisecond time scales.
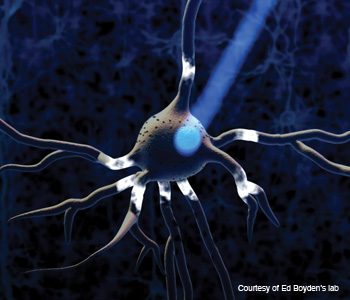 |
| A tiny light on an individual neurone causes a change in the behaviour of the neurone. |
While this technique offers incredible time resolution, however it's incredibly invasive (you have to insert the fibre-optic fibres into the brain of the animal) and incredibly difficult to get right (you need to make sure the fibres are actually lighting up the neurones you want them to). This creates a huge problem if you want to target neurones deep within the brain, as you need access to those incredibly tricky to reach parts of the brain.
Chemogenetics
Another cool way scientists have been able to manipulate the brains of animals using genetics have involved the use of chemicals, or toxins to be more specific. Researchers have used the Cre-lox system to insert the gene for various toxins eg a fragment of the diptheria toxin into specific neurones in order to kill those cells and observe the effects this has on the mice. Although a bit crude, this can help to identify the brain regions involved in all different types of behaviours, and could help us understand how the brain reacts to and compensates for the loss of specific regions of the brain eg after stroke, dementia, trauma.
Electrogenetics
This technique doesn't necessarily allow researchers to directly manipulate neurones, but it does allow us to watch the activity of specific cells in real time. By inserting genes that express proteins that emit a flourescent light when they sense changes in voltage (indicating electric activity in the neurone) we can easily see each neurone light up individually and trace which other neurones are activated or de-activated by that neurone.
This video below used this technique to show how individual neurones in the auditory cortex are firing in response to sound.
Along the same lines, you can insert specific genes across the entire brain that randomly express a different ratio of red, green and blue fluorescent proteins. That means that each individual neurone has a unique colour, which gives rise to the incredible 'brainbow' images seen above.
Magnetogenetics
This very new method of manipulating neurones involves inserting a gene for an ion channel protein that is activated by magnetic fields you can instantly turn neurones on or off or block anything else from activating it (ie other neurones) while the animal is relatively free to move around and act as normal, without needing a bunch of fibre optic cables delicately positioned in their brains. The fact you don't need to worry about exact positioning of any equipment means that much deeper brain regions or much more specific neurones can be manipulated than with optogenetics. There may also be potential for more analogue control of activity by using a weaker magnetic field that only slightly affects the activity of the neurones being manipulated. However some neurones are already sensitive to magnetic fields, birds and other animals can use them to navigate by, for example.
 |
| I couldn't not use this picture once I found it! |
Ultrasound-ogenetics?
Probably better named audiogenetics (although we can't hear ultrasound...), there is also some very early research into making specific neurones sensitive to ultrasound. This would have many of the benefits of magnetogenetics and both could be the future of genetics based neuroscientific research in animals.
Clearly, there are a number of new and emerging ways in which researchers may be able to investigate the role of very specific neurones in the brain. While some may be initially put off by the idea of manipulating the brains of animals, by understanding these very specific roles, we may eventually reach a more complete understanding of how the brain works and how to fix it when it goes wrong.
Comments
Post a Comment